 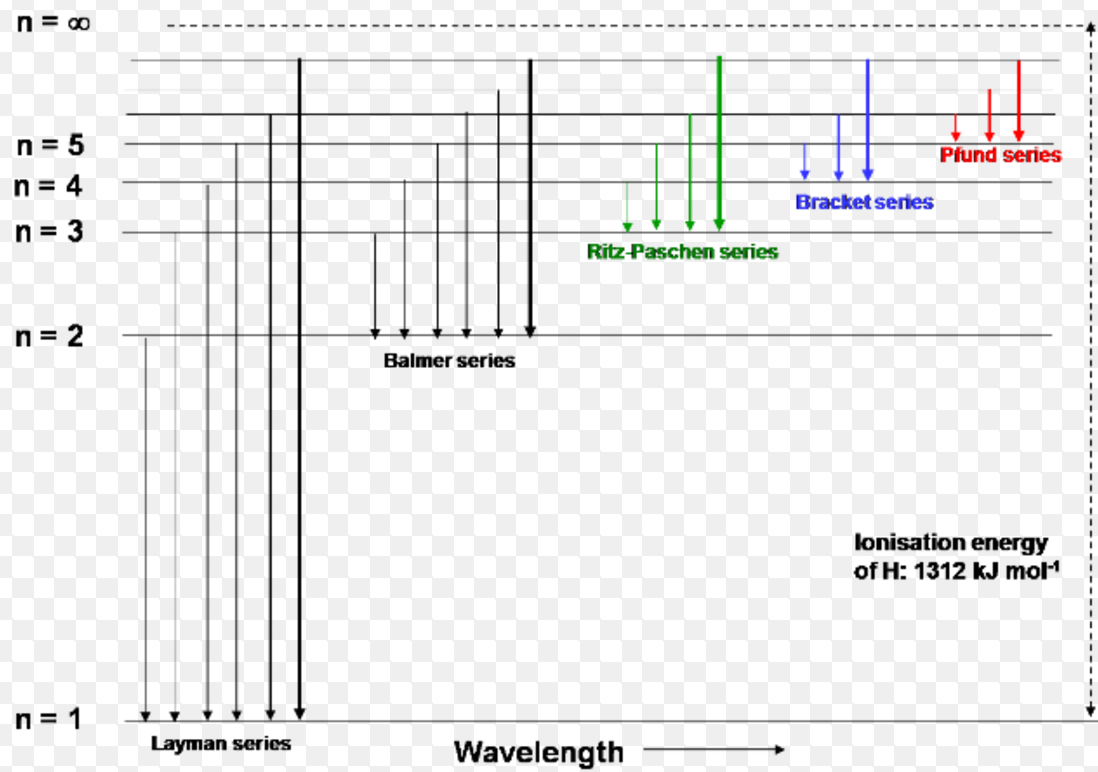
Propose How This Might Be Done, And What The Absorption. Nist atomic spectra database, gfall.dat file from kurucz cd. These frequencies of photon of light are then missing in the spectrum. List The Spectral Lines For Hydrogen Gas By Color And Corresponding Wavelength. Use colored pencils to color the hydrogen and boron spectral lines within their respective spectra in model 2. Scientists can also record the absorption spectra for elements. The Characteristic Pattern Of Molecular Emission Produced By Boron Oxides (Bo 2 ) (Ao Et Al.Ģ017) in vapor phase was visible in both pristine and fluoropolymer coated. Bohr's model of hydrogen is based on the nonclassical assumption that electrons travel in specific shells, or orbits, around the nucleus. Scientists can also record the absorption spectra for. When white light is shone through a gaseous form of an element, the electrons absorb photons of light of specific energy. Sodium (Na), Hydrogen (H), And Helium (He). Source: Use colored pencils to color the hydrogen and boron spectral lines within their respective spectra in model 2. Propose how this might be done, and what the absorption. The three atomic emission spectra for boron can be shown on graph bellow: Source: The characteristic pattern of molecular emission produced by boron oxides (bo 2 ) (ao et al. Source: The absorption spectrum of hydrogen shows the results of this interaction. Source: The three atomic emission spectra for boron can be shown on graph bellow: Model 2 shows the emission spectra for hydrogen and boron. Source: In the visible part of the spectrum, hydrogen absorbs light with wavelengths of 410 nm (violet), 434 nm (blue), 486. The absorption spectrum of hydrogen shows the results of this interaction. The characteristic pattern of molecular emission produced by boron oxides (bo 2 ) (ao et al. Source: Scientists can also record the absorption spectra for elements. In the visible part of the spectrum, hydrogen absorbs light with wavelengths of 410 nm (violet), 434 nm (blue), 486. Source: The characteristic pattern of molecular emission produced by boron oxides (bo 2 ) (ao et al. According to the bohr model, the wavelength of the light emitted by a hydrogen atom when the electron falls from a high energy (n = 4) orbit into a lower energy (n = 2) orbit.substituting the. Source: Bohr's model calculated the following. Model 2 shows the emission spectra for hydrogen and boron. (PDF) High resolution Fourier transform spectroscopy of H_2 IR emission from Scientists can also record the absorption spectra for. Sodium (na), hydrogen (h), and helium (he). Model 2 Shows The Emission Spectra For Hydrogen And Boron. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |



 RSS Feed
RSS Feed
